blog
October 14, 2010 | Source: Admin User, Inc.
Webcast on Automated Microbial Testing -- Audience Polling Responses
POSTED BY Admin User | 4 minute read
October 14, 2010 | Source: Admin User, Inc.
POSTED BY Admin User | 4 minute read
A few weeks back, we hosted a webinar through American Pharma Manufacturing entitled, “Bringing your Micro QC lab into the 21st century: The Role of Automated Methods”. The webinar explored where companies are today in their use of rapid microbial detection automation, and how available technologies can help them move forward. Thanks to our speakers, Bikash Chatterjee of Pharmatech Associates, Diluks De Silva of Novartis, and our own David Jones. If you haven’t watched the event, it is worth the 45 minutes.
The session was interactive with polling questions to the audience about their current practices in the micro QC lab. The answers provided to our unscientific poll by the 120 attendees offers a window into current processes and roadblocks in pharmaceutical quality control in the micro QC lab.
Question 1:

The results of this first poll were very encouraging. The bulk of the respondents answered that they are efficient and have some level of automation. Companies realize the importance of automation and have taken steps to bring devices to their micro QC Lab that help to streamline their processes. The 25% who are still in the 19th century should be applauded for their honesty. Perhaps they are with businesses that would be considered late adopters, although the next question may offer some insight on why only 11% answered that they are highly advanced.
Question #2
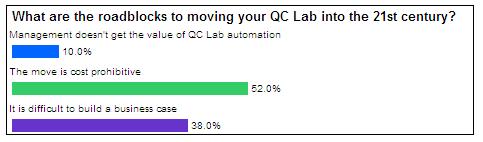
More than half the respondents answered that cost was a major roadblock to moving forward with additional automation. One could argue that cost along with the difficulty of building a business case are intertwined. Companies that build a compelling business case for automation of the micro QC lab would likely prove an ROI that mitigates the cost issue.
These two challenges are fairly common with companies to whom we speak. We recently wrote an executive whitepaper with some guidelines to help businesses build their business case for automated rapid detection. Only 10% of the polling responses indicate issues convincing management on the value of QC lab automation. I would say that is good news, and also an area where a business case can help
Question #3
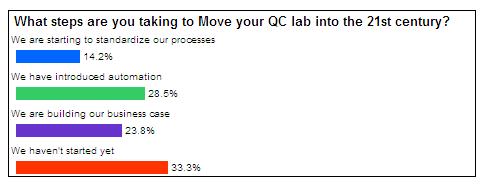
This final question showed that most companies are on a path towards automation in the micro QC lab, although at different stages. Whether they are building their business cases, standardizing their processes in preparation to automate them, or introducing automation, over 66% are on the path to the 21st century. Unfortunately, over a third of the respondents to this question have started. Building their business case may be the best place to begin their journey.
Stay tuned for future events like these.
Mark Severns
Marketing Manager
Rapid Micro Biosystems