blog
May 29, 2018 | Source: Rapid Micro Biosystems, Inc.
Growth Direct™ brings Industry 4.0 to the QC Micro Lab
POSTED BY Rapid Micro Biosystems | 4 minute read
May 29, 2018 | Source: Rapid Micro Biosystems, Inc.
POSTED BY Rapid Micro Biosystems | 4 minute read
Industry 4.0 is the concept of connecting human resources, data, and machines into one virtual network. This concept has been around for several years, but recently, Industry 4.0 has extended to Pharma. Pharma 4.0 brings the promise of improved “quality, productivity and lead times through inter-connectivity and automation[i].” ISPE has formed a special interest group around Pharma 4.0, and they advocate that Pharma 4.0 should include design features such as “data integrity by design” that will complement the Quality By Design processes that many firms are already using.
Additionally, the “Human Factor” has been labelled by some as the most powerful concept of Pharma 4.0. True Pharma 4.0 solutions are technological solutions that will make available companies’ greatest assets: their human workforce. These 4.0 solutions will “minimize humans-to-process interaction and in turn reduce human error, operator-to-operator variability and human related contamination steps[i]”. By liberating workforces from non-value-added steps, Pharma 4.0 solutions will allow workers to focus on “real value-added activities where the creativity and problem-solving capabilities of the most advanced machine in the world – the human brain – can be utilized to their fullest potential[ii].”
Growth Direct and Pharma 4.0: Improved Quality, Productivity and Leadtime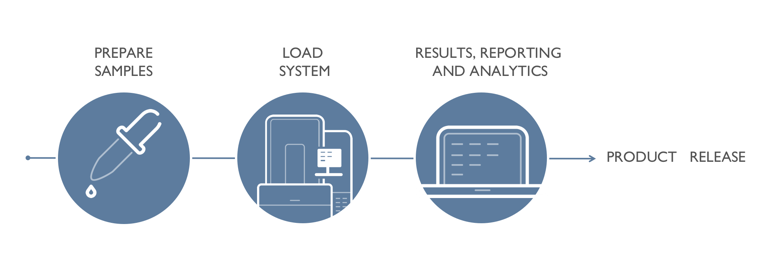
Growth Direct Improves Quality
The Growth Direct was specifically designed to enhance labs’ data integrity and sample safety. It tracks samples via unique barcodes and has secure software that was engineered to be compliant with 21 CFR Part 11. All data resides in an encrypted database in its permanent form for the lifetime of the record. Each step requires unique authentication, and the final result is easily accessible to authorized users. For more information on the Growth Direct’s data integrity features, click here.
Growth Direct Improves Productivity
The Growth Direct system automates traditional QC microbiological incubation and enumeration processes. It minimizes human errors due to misplacing or mislabeling plates, miscounting, and transcription errors. The Growth Direct liberates people in QC Micro labs to perform true value-added tasks like release testing and productivity improvements that make manufacturing processes safer and more efficient. For more information, sign up for an analysis of your lab.
Growth Direct Improves Leadtime
The Growth Direct system detects microbial growth within hours and yields final results in approximately half the time of the traditional methods. Your organization could save days of manufacturing or warehousing costs by implementing the Growth Direct in your QC Micro lab. For more information, sign up for a Growth Direct Workshop.
[i] Chatterjee B. “Pharma 4.0: A New Framework & Process For Digital Quality Management,”