blog
March 28, 2024 | Source: Rapid Micro Biosystems, Inc.
The Future of Bioburden Testing is Automation
POSTED BY Rapid Micro Biosystems | 4 minute read
March 28, 2024 | Source: Rapid Micro Biosystems, Inc.
POSTED BY Rapid Micro Biosystems | 4 minute read
Rapidly wishing away the painful waiting for QC test results? In microbial tests such as bioburden, harnessing the power of technology can help.
Bioburden is a critical step to microbiological safety, ensuring that the product is clean and free of bacteria during the manufacturing process. It gives data on both the expected microbial load of the product and the presence of specific organisms, enabling intervention before a contaminated product is released to market.
As any microbiologist knows, this test is crucial yet tedious, with pulling, shifting, and recording samples multiple times for a single batch. The repercussions of errors in testing can range from batch losses and product release delays to product recalls and facility shutdowns.
The three main test options for colony enumeration, as described in USP <61> and <1119.1>, are membrane filtration, pour-plate, and the surface-spread method. Membrane filtration is an established and reliable method for fast, flexible testing, especially for samples with low microbial loads or when specific organisms need to be recovered, isolated, and identified.
Each of these techniques relies on visual plate counting as the primary method for microbiological analysis. However, counting visible colony growth by eye brings a few major challenges:
In addition, even with gowning, gloving, and other proper cleanroom procedures, humans are the most significant source of contamination. The handling of samples when moved in and out of incubators for testing, when performing interim counts, and when making transfers increases the chances that the sample will be inadvertently compromised with secondary contamination.
Benefits of Automation
Addressing these challenges, the Growth Direct® System is an RMM (Rapid Microbial Method) that improves on traditional membrane filtration, delivering increased accuracy, a faster time to result, enhanced data integrity compliance, and more control over the manufacturing process.
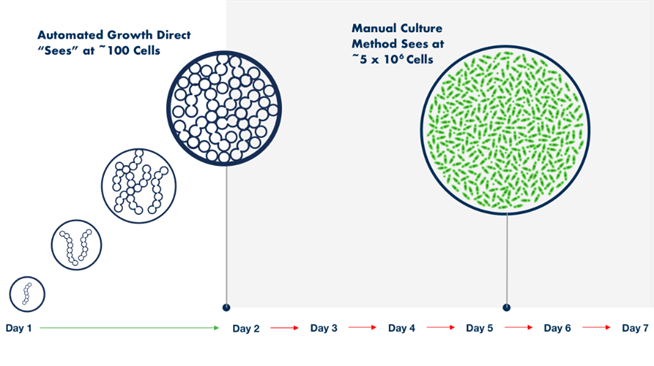
A key differentiator and advantage of the Growth Direct® over traditional membrane filtration is the platform technology’s ability to detect colonies several generations before they become visible to the naked eye (Figure 1). This means faster product release, with traditional testing timeframes shortened by up to 50%. It also enables faster remediation in the case of a contamination event through quicker decision making, investigation, and root cause findings.
 The system is designed to adhere closely to the compendial methods outlined in USP <61> and continues to align with the new USP <1119.1>, meaning seamless compliance with current and future regulations. It meets the dual-read requirement from regulators while vastly reducing risk from human error in handling, counting, and data entry.
The system is designed to adhere closely to the compendial methods outlined in USP <61> and continues to align with the new USP <1119.1>, meaning seamless compliance with current and future regulations. It meets the dual-read requirement from regulators while vastly reducing risk from human error in handling, counting, and data entry.
With an increased reliance on pharmaceuticals for the livelihood of patients worldwide, automation of microbial testing is more important than ever. Pharmaceutical manufacturers must keep up with regulatory requirements and product supply demands while staying on top of the major risks in traditional microbial quality control processes. The technology to do so is already here; contact us to learn more.