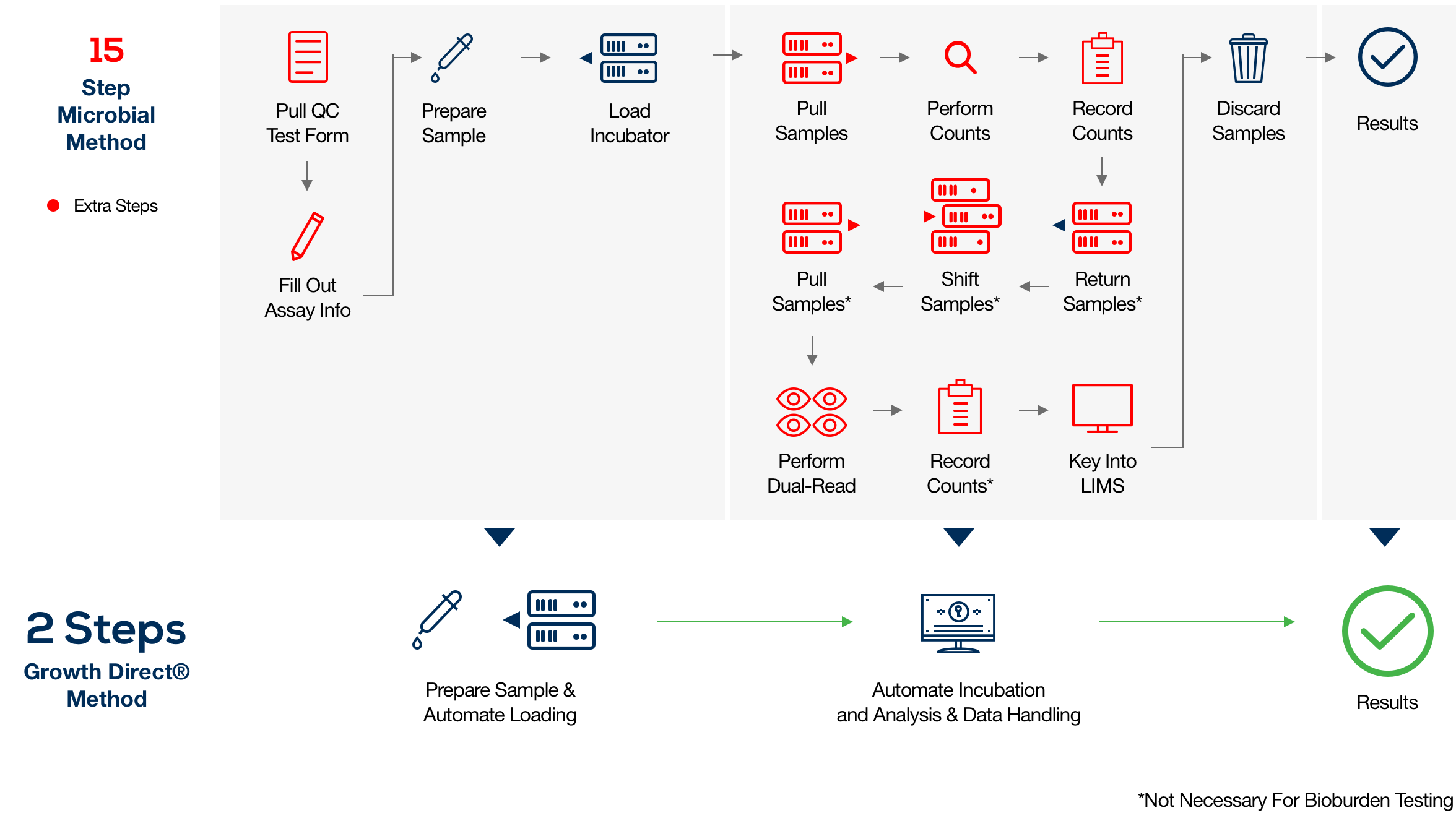
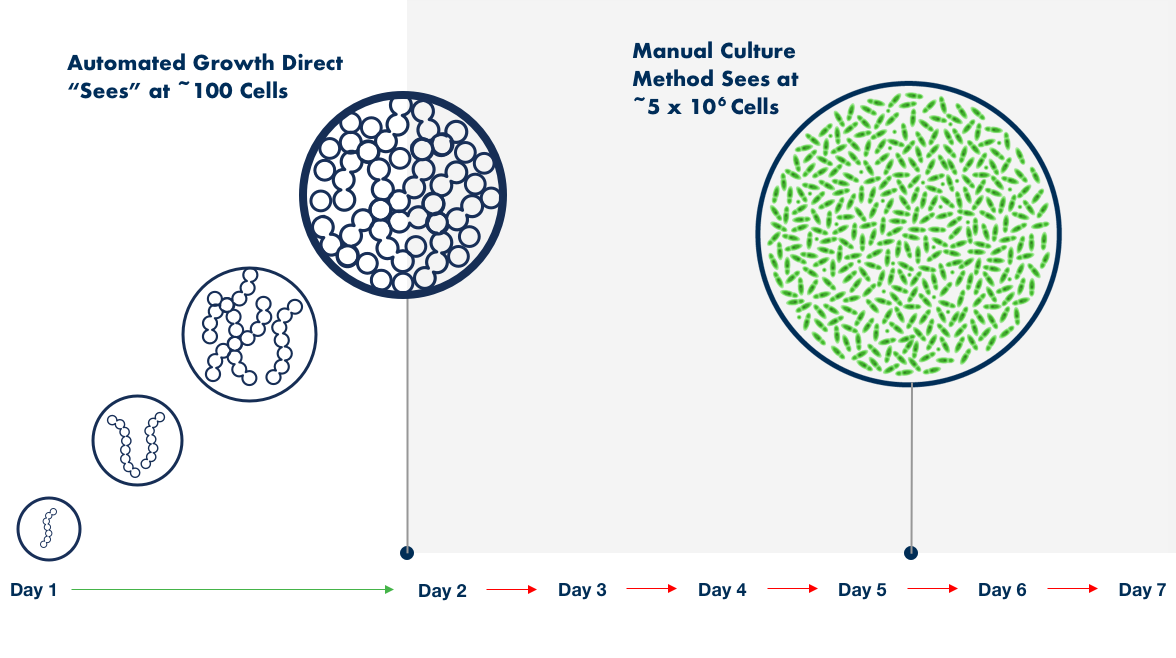
Bioburden testing is a critical quality control (QC) step for contamination detection, but the traditional compendium method can fall short of today's speed and safety demands. Sample enumeration, tracking, and reporting are typically subjective, time-consuming manual processes prone to human error.
The Growth Direct® System overcomes these challenges with novel growth-based technology that features advanced robotic automation, powerful optical imaging, algorithmic vision analysis, and data management. The result is fast, accurate enumeration and digitization that can boost productivity in your microbial QC lab while eliminating manual sample analysis and recordkeeping. Click here to learn more in our *NEW* bioburden testing eBook and Infographic.
.jpg)