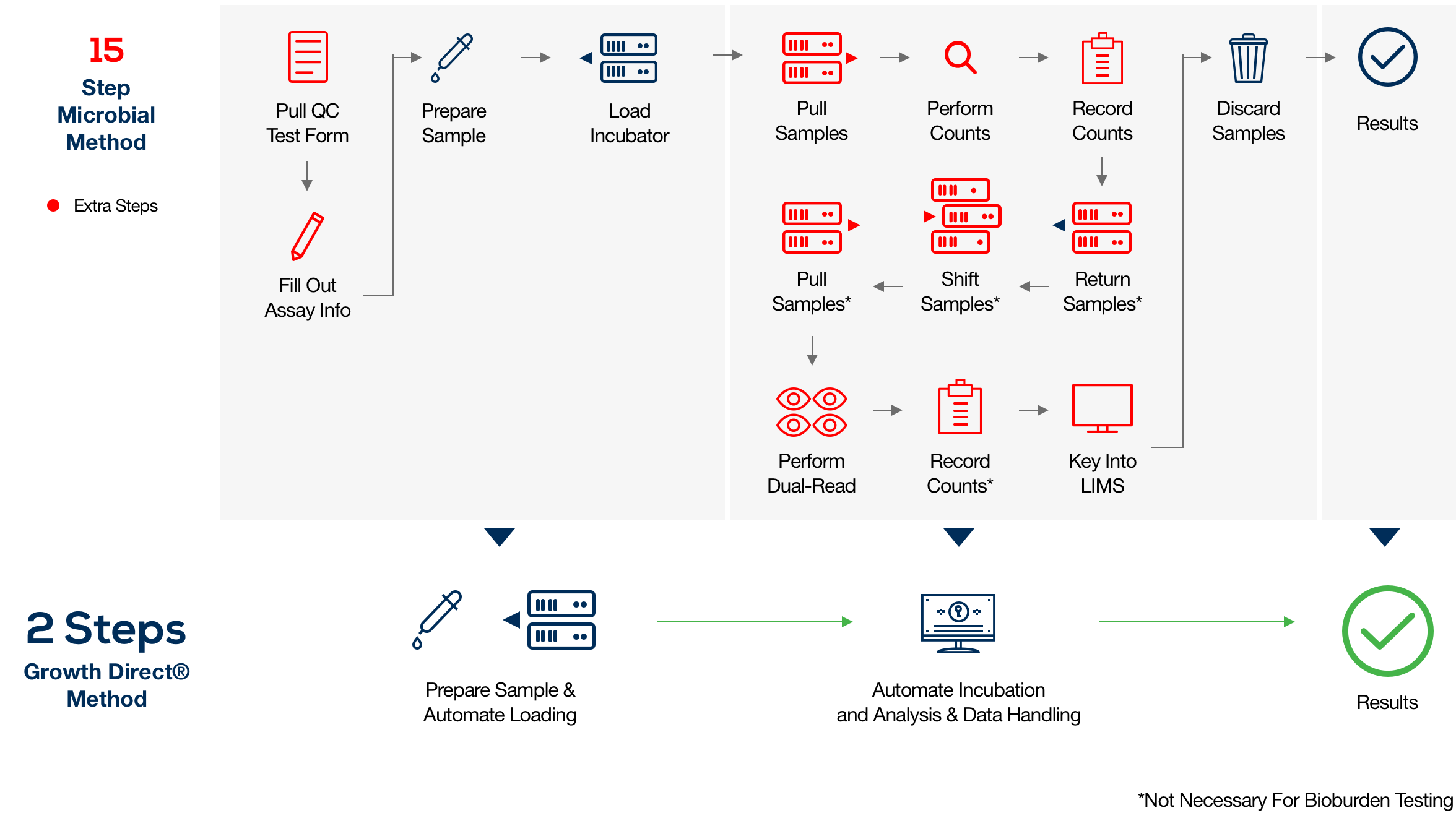
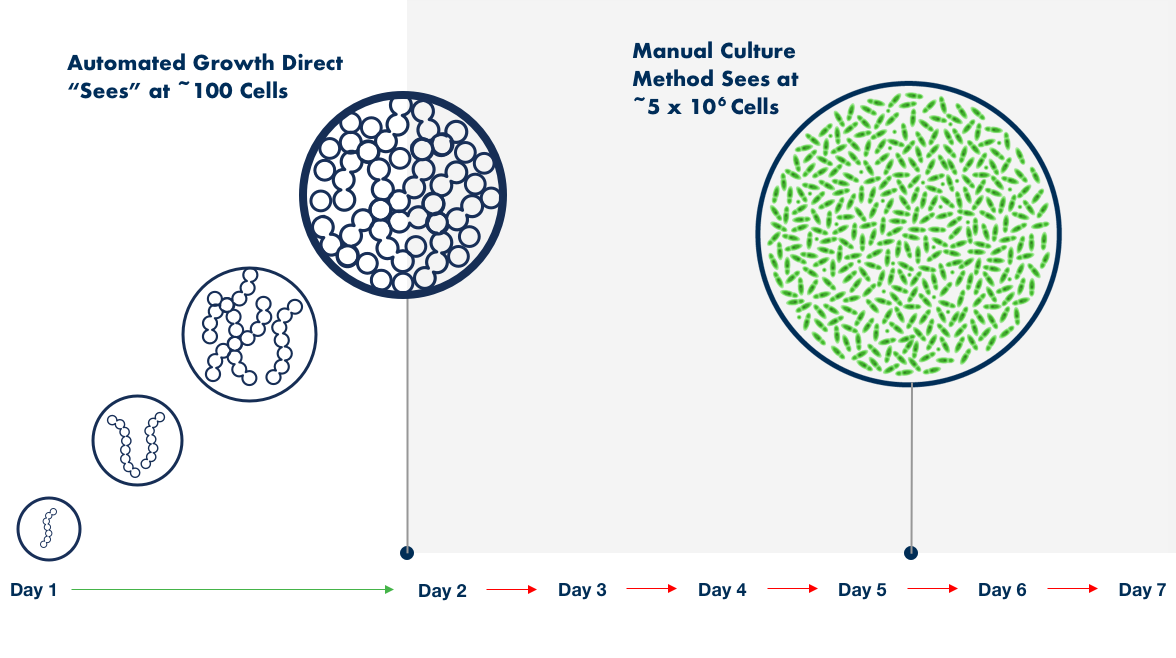
The sheer volume of water that must be tested and analyzed for quality control (QC) can pose serious challenges in QC labs—particularly when sample enumeration, tracking, and reporting are based on manual processes that can be highly subjective, time-consuming, and prone to human error.
The Growth Direct® System overcomes these challenges with novel growth-based technology that features advanced robotic automation, powerful optical imaging, algorithmic vision analysis, and data management. The result is fast, accurate enumeration and digitization that can boost productivity in your quality control microbiology lab, eliminating manual microbiological analysis of water and recordkeeping while upholding cGMP principles.
.jpg)