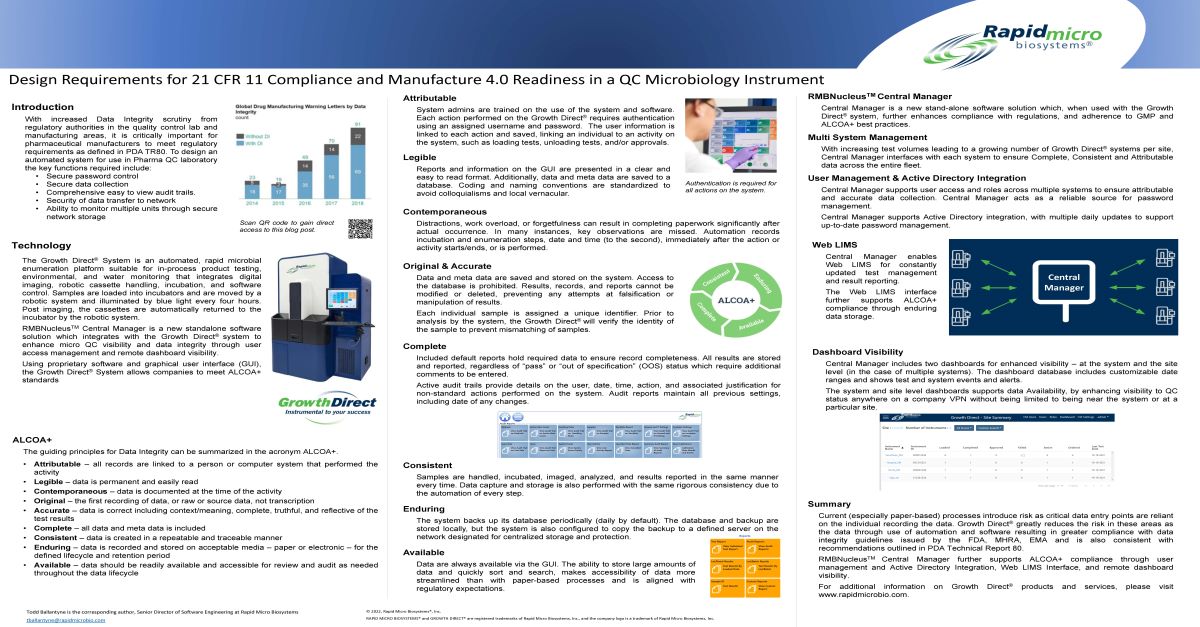
2026 Events, Conferences, and Webinars
American Biomanufacturing Summit — San Francisco, US
April 14–15, 2026
Swiss Cleanroom Conference — Pratteln, Switzerland
April 27, 2026
Cell & Gene Meeting on the Med — Rome, Italy
April 28–30, 2026
GENAP 2026 — Vienna, Austria
May 5–6, 2026
Pharma Manufacturing World Summit — Boston, US
May 18–20, 2026
Growth Direct Day 2026 — Thousand Oaks, CA, USA
June 10–11, 2026
PDA Good Aseptic Manufacturing Conference 2026 — Frankfurt, Germany
June 11–12, 2026
IMAPAC Cell and Gene Therapy World Asia — Singapore, Singapore
June 24–25, 2026
A3P Microbiology Conference — Strasbourg, France
June 24–25, 2025
BPI International — Boston, US
September 22–25, 2026
Farmaforum — Madrid, Spain
September 22–23, 2026
Aseptikon 2026 — Mannheim, Germany
October 7-8, 2026
A3P International Congress Sterile Manufacturing & ICHQ10 — Biarritz, France
October 7-8, 2026
PDA Aseptic Manufacturing Excellence Conference — Melbourne, Australia
October 13–14, 2026
PDA Pharmaceutical Microbiology Conference — Washington, D.C., US
October 26–28, 2026
Aseptic Processing Summit — La Jolla, US
October 27–28, 2026
Pharma Microbiology Congress (PMC) — Florence, Italy
October 29, 2026
Pharmalab — Darmstadt, Germany
November 23–25, 2026
Past events
GMP-PharmaCongress 2026 — Wiesbaden, Germany
March 24–25, 2026
Korea Lab — Seoul, South Korea
March 31–April 3, 2026
IMAPAC Biologics Nordics — Stockholm, Sweden
March 18-19, 2026
WEBINAR: Validating Automated Alternative Rapid Sterility Methods - online
February 10, 2026, 12 PM PT, 3 PM ET
A3P Spain - Forum Microbiology — Madrid, Spain
February 24, 2026
.jpg)